
They have one electron found in their outermost shell. Francium, however, is a laboratory-produced element and only minute quantities have been made, so for all practical purposes, the most reactive metal is cesium. 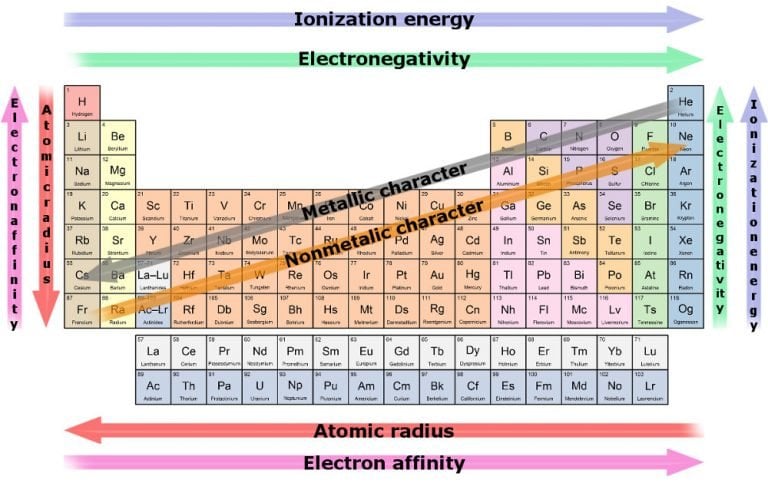
They are known as group $1$ elements and are found in the s-block of the periodic table. The most reactive metal on the periodic table is francium. Elements at the top of the reactivity series are the most reactive, while those at the bottom are the least reactive. The greater the shielding, the greater the ability to lose electrons. Metals become less likely to oxidize, tarnish, or corrode. Atoms lose electrons less easily to form cations. We have to know that members present in alkali metal family are potassium $\left( K \right)$, sodium $\left( \right)$. In general, the bigger the atom, the greater the ability to lose electrons. The most reactive metals are on the bottom left side of the periodic table. The reason why they are highly reactive is because of the electronic configuration. The most reactive metal is cesium, while the most reactive nonmetal is fluorine. We can say that the family which has the most reactive metals in the periodic table is alkali metals. As such, as you increment the size of atoms of metals in a group, their reactivity likewise rises. These electrons are simpler to eliminate, so the atoms promptly form chemical bonds. These elements share some properties with both metals and nonmetals. Low-reactivity metals are safer to handle than high-reactivity ones. Reactivity refers to how easily a substance reacts chemically with other substances. As you drop down a column or group of the periodic table, there would be an increase in size of the atomic radius.įor the metals, this implies the valence electrons turn out to be farther away from the nucleus which is positively-charged. Familiar metals also are less reactive than most other metals. A component that is exceptionally electronegative, like fluorine, has an incredibly high attraction for holding electrons.Ĭomponents at the far edge of the range, like exceptionally reaction metals cesium and francium, promptly form chemical bonds with electronegative particles. This above image clearly shows you where are Alkali metals located on the Periodic table. Alkali metals include: Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs) and Francium (Fr). We have to know that Reactivity is a proportion of how possible a chemical species is to partake in chemical reactions to produce chemical bonds. Alkali metals are located in group 1 on the left side of the Periodic table. The easier it is for a metal to lose electrons, the more reactive it is. Based on the periodic trends in the last 3 sections, this means that they are usually bigger, more likely to lose electrons, and less likely to gain electrons, than the non-metals. The arrangement is a chart that rundowns components as per how promptly the metals displaces hydrogen in chemical reaction. Remember that metals are on the left and bottom of the periodic table. Hint: We have to know that one can utilize the reactivity of metals series to foresee which metal will be the most reactive and to look at the reactivity of various metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
